By Dr. Patrick Howson
Along with collaboration and a commitment to supporting our people, scientific integrity is one of the three core pillars that guide Atuka as an organization. From an unbending commitment to animal welfare to our absolute dedication to quality in ensuring reliable and repeatable data, all our scientific work is governed by rigorous, internal quality control systems. All our studies abide to a self-established quality system that describes the processes and conditions under which studies are planned, performed, monitored, recorded, archived and reported. Underpinning this system is a suite of Standard Operating Procedures that defines these processes. Ultimately, the Atuka quality system ensures that the services we provide are of the highest standard, are ethical, and that data generated are reliable, accurate and precise.
Improving the quality of preclinical data is an important and necessary process. The success rate in the discovery of novel therapeutics has been declining for decades and there are many reasons why this has been happening, such as tackling more complex disorders, a focus on developing drugs that treat the underlying disease rather than the symptoms, and unreliable translation from preclinical efficacy to clinical benefit.
One key reason, however, for declining success is a decrease in the repeatability and robustness of studies. It has been reported that:
- 70% of studies, repeated in a different lab did not replicate the initial finding
- 50% of studies, repeated in the same lab did not replicate the initial finding
- A large biotech company repeated 53 “landmark studies” and could only replicate 6 (11%) of them
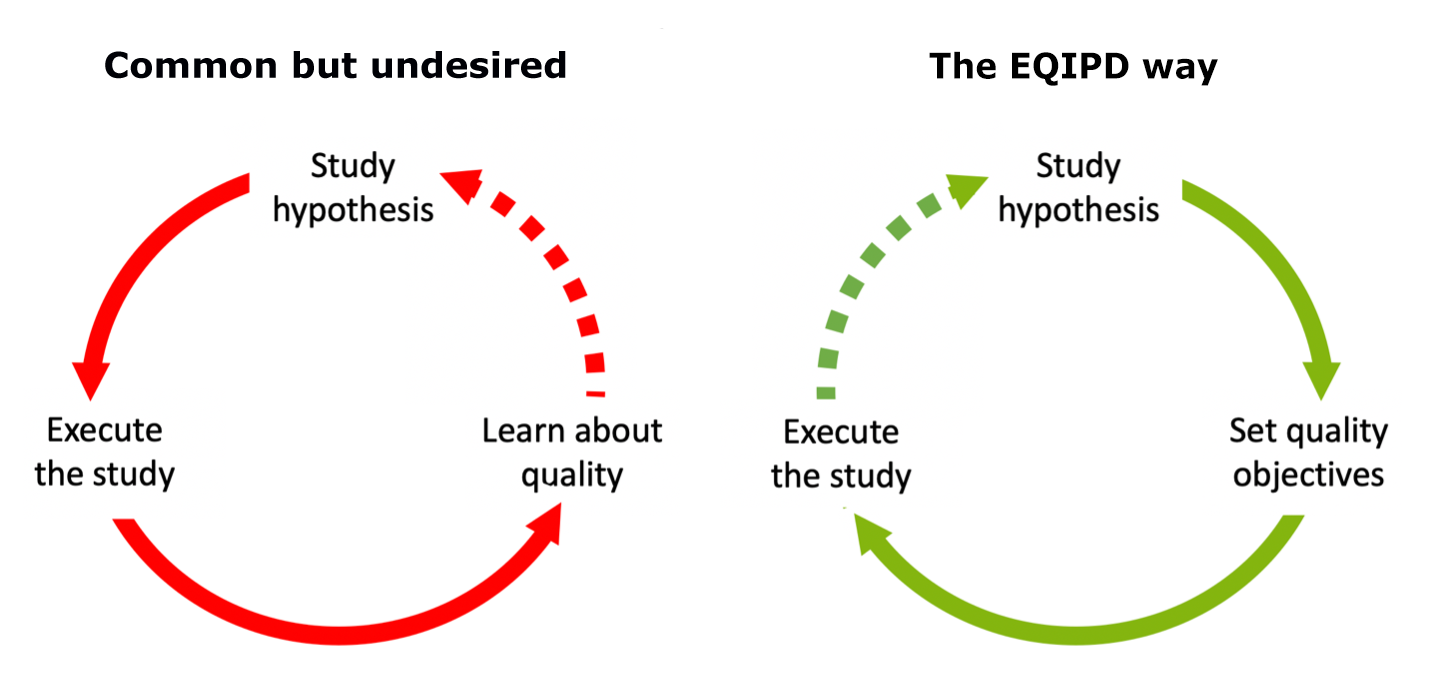
There are likely many reasons behind this lack of reproducibility, but one key mechanism for improving reproducibility is enhancing the rigorousness of research, i.e., having a quality system in place that minimizes biases and ensures a high degree of standardization across the procedures that underpin scientific studies. The quality systems at Atuka have thus been designed with this purpose in mind.
There are several quality systems governing research and analysis in a preclinical setting. Two of the better-known systems are Good Laboratory Practice (GLP), which is used in preclinical safety and toxicology studies, and ISO17025, which focuses on standardization of a process that is continually repeated, for example testing of microbial levels in food. These systems have been designed to address specific issues, namely minimizing the risk of a dangerous compound being tested in humans (GLP) and minimizing the risk of a released product being out of a pre-specified specification (ISO17025). However, these quality systems were not designed to minimize the risk of generating unreliable, and thus unrepeatable, efficacy data whilst not restricting the freedom to explore scientific questions.
To address this gap, a new quality system has been developed called EQIPD (Enhancing Quality in Preclinical Data). Initially supported by the European Union’s Innovative Medicines Initiative, EQIPD addresses one of the central challenges of preclinical biomedical research—facilitating the reproducibility of results across a wide range of laboratory settings. Thereby ensuring that all of us, whether in public institutions or private industry, are working toward common goals supported by the best possible data.
Atuka’s commitment to the highest quality of scientific research means continually appraising and improving our quality systems and always going the extra mile to do better. Which is why we are pleased to announce that Atuka has begun the process of aligning our quality systems with the EQIPD guidelines. As the quality system currently in place at Atuka was designed to minimize errors and biases in the data we generate, it is already well aligned with many of the core concepts of EQIPD.
Over the coming months, as our alignment with EQIPD proceeds, we will be taking a closer look in this space at EQIPD to better explain, and understand, the core concepts of the quality system, and highlight how Atuka minimizes the risk of generating biased data in ways that align with its core concepts.
🔗 Learn more in this “Introduction to the EQIPD quality system”.